CASE STUDY: Babyation Breast Pump
From Concept to TIME’s Product of the Year in 24 Months
How we helped revolutionize breast pump technology with full-stack product development and rapid time to market.

A Medical Device for Modern Mothers
Services
Hardware Engineering, Firmware Development, App Development, Cloud Architecture, FDA Regulatory Compliance
Project Timeline
18 months to FDA approval
24 active months to market launch
Tech Stack
Embedded Systems, iOS/Android, AWS Cloud Infrastructure, BLE/Wi-Fi Connectivity, HIPAA-Compliant Security
“Everybody else they went to said ‘we can build your hardware but not your cloud,’ or ‘we can build your app but not your hardware.’ We said ‘we can build everything for you, plus we can help you navigate the FDA.”
Jon, Systematic Consulting Group
Sam’s Challenge
Sam had just given birth when she discovered what millions of working mothers already knew: breast pump technology was stuck in the 1990s. The devices were loud, tethered to wall outlets, and impossible to use discreetly at work. She and her husband Jared had a vision for a smart, quiet, portable pump that would seamlessly integrate into modern life.
But every development firm they approached could only build pieces of the solution. One could handle hardware but not software. Another knew apps but not FDA compliance. The few claiming comprehensive capabilities quoted 3-4 year timelines at budgets that would sink their startup.
We were able to offer complete end-to-end development—hardware, firmware, app, cloud, and FDA approval—in months and not years.
Read on to find out how we delivered on that promise.

Define & Discovery
Strategic Roadmapping Before Development
The project began with a Phase 0 consultation. Rather than rushing to build, the team mapped the entire journey from concept to FDA approval. This strategic planning revealed opportunities to compress timelines and reduce costs that would have been invisible once development started.
We were ultimately tasked with creating Babyation’s complete technology ecosystem: the intelligent pump hardware, the mobile app for discrete control, the cloud infrastructure for data management, and the comprehensive documentation required for FDA approval.
Defining the Technical Revolution
Discovery began with competitive analysis and user empathy mapping. Nearly every pump on the market required users to repeatedly press buttons to adjust settings—hardly discrete in a workplace setting. Babyation would feature an intuitive display with app-based control, allowing mothers to adjust settings privately from their phones.
The team identified key innovations that would differentiate Babyation:
- Smart tracking: Automatic logging of pumping sessions and milk production
- Extended battery life: Multi-day operation versus competitors’ few hours
- Silent operation: Discrete enough for conference calls
- App connectivity: Seamless app connectivity over BLE
To achieve FDA approval in record time, we implemented end-to-end requirement traceability from day one. Every design decision, test result, and safety protocol was documented with the rigor FDA reviewers demand.
Architect & Design
Component Selection and Architecture
Following discovery we began component analysis, focusing on parts that would deliver medical-grade performance in a consumer-friendly package. The team worked hand-in-hand with mechanical design contractors to ensure perfect integration.
Key decisions made during this phase:
- Processor selection for real-time performance and power efficiency
- Battery technology enabling multi-day operation
- BLE/Wi-Fi hybrid connectivity for reliable app communication
- Medical-grade pump mechanisms with patented vacuum boost technology HIPAA-compliant encryption for data security

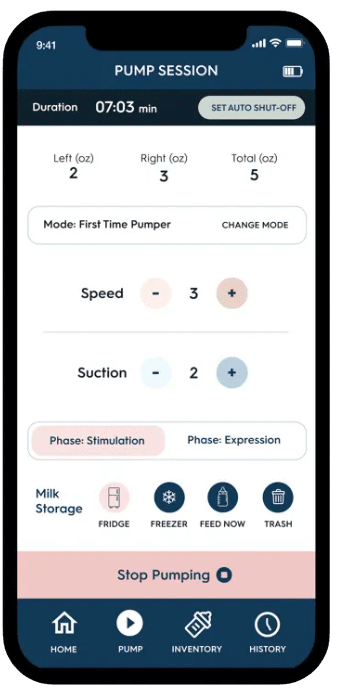
Develop & Iterate
Parallel Development Tracks
With architecture defined, four specialized teams worked in synchronized sprints:
Hardware Team: Developed the smart pump system with revolutionary battery life and quiet operation. The custom motor and pump system delivered hospital-grade suction in a portable form factor.
- Firmware Team: Developed software and algorithms running on the hardware
- Software Team: Built the intuitive iOS and Android apps with real-time device control, automatic session logging, and milk inventory tracking. The interface was designed for one-handed operation—critical for multitasking mothers.
- Cloud Team: Created secure, HIPAA-compliant infrastructure for data synchronization and storage. Every piece of health data was encrypted end-to-end.
Daily integration meetings ensured perfect harmony between all systems—a key factor in achieving the aggressive timeline.
Deploy & Optimize
Prototype to Production
Within twelve months, we delivered working prototypes that validated the complete user experience. Sam could pump discreetly at work, controlling everything from her phone while the device automatically logged production data.
The team then shifted focus to FDA requirements:
Electromagnetic interference testing
Human safety validation
Biocompatibility verification
Security and privacy audits
End-to-end requirement documentation
SCG’s meticulous approach to documentation—built throughout development rather than retrofitted—proved crucial. The FDA approved Babyation on the first submission with zero modifications required, an almost unheard-of achievement.


The Results
Systematic Consulting Group transformed Sam and Jared’s vision into market reality in a little under two years—75% faster than industry average.
When mothers use Babyation, they experience technology that actually fulfills their needs. The pump operates quietly enough for video calls, runs for days on a single charge, and tracks everything automatically. No more logbooks, no more dead batteries, no more embarrassing mechanical sounds in the office.
The market response was extraordinary. TIME Magazine named Babyation one of the year’s best inventions. The product that started as a frustrated mother’s idea became a category-defining innovation that’s transforming how women balance motherhood and career.
On top of everything else, we provided full documentation at the end of the project that enabled Babyation to successfully transition to in-house operations, with the team maintaining a consulting relationship for ongoing optimizations.
Today, Babyation stands as proof that medical device innovation doesn’t require years of development. With the right partner, revolutionary products can reach market in months, not years.
Ready to bring your idea to life? Schedule a consultation today.